
Fars Plus magazine; Mohammad Taghi Khorsandi: The production of 50 million doses of vaccine by the end of September is not something that no Iranian has heard of. The promise that the informant, the head of the executive staff of the Imam’s order, repeated many times and even insisted on its implementation until the end of July. But now, there are only a few days left until the end of September and the number of vaccines has reached 10 million doses. Although the Barakat 2 line has recently entered the production circuit, it is practically unlikely that the number of vaccines produced will exceed 15 million doses. What caused this promise to be delayed? If only US sanctions were an obstacle, if there were no sanctions in these 40 years, then why didn’t the executive staff anticipate it?
Sanctions in the moment!
Barakat vaccine reached the stage of human testing on January 30, 2017. The first US response was clear: the Barakat Foundation boycott in less than two weeks. The vaccine, the Corona vaccine, was not something the United States joked about. Playing with people’s health has always been the most important American tool for creating dissatisfaction in Iran. On the other hand, the entry of a new player into the vaccine market could seriously jeopardize the technological monopoly and high profitability of American companies.
But sanctions in Iran have a dual effect; It is both an obstacle to progress and a factor in great progress. When we become a boycott, we have to stand on our own two feet. Of course, they try not to let go, but wherever we want, we have succeeded. The story of the vaccine was the same.
Two weeks after the clinical trial of the Barakat Institute vaccine began, the United States boycotted it
Everything from scratch
Given the importance of the time factor in the fight against corona, Barakat put mass production of the vaccine on its agenda from the day its initial tests were successful. The success of this work required the launch of a vaccine production line from scratch. Because Iran needed at least 160 million doses of vaccine and none of the existing infrastructure in the country could meet it; Neither laboratories nor production lines.
The main steps in producing the active ingredient of the corona vaccine include culturing the cell, then replicating the virus, and finally killing the virus and filtering it. This technology was what introduced Iran to the list of 6 vaccinating countries in the world. But it also required advanced laboratories with a high level of biosafety. These labs are classified into 4 levels known as BSL 1-4 (BioSafety Level 1 to 4). It is a suitable laboratory for the BSL4 coronavirus, but due to US sanctions, it was almost impossible for Iran to access it.
The best active laboratory in Iran was a BSL2 laboratory that was not safe for the Corona virus. The largest BSL3 laboratory in the Middle East in Mashhad, with 92% progress, had been stopped for more than 12 years and its equipment was being destroyed. For this reason, the construction of a modern, well-equipped and large BSL3 laboratory was on the agenda from the very beginning of the research, so that it could mass-produce cells in accordance with the needs of industrial production lines.

One of the infrastructures needed to produce the Barakat vaccine was an advanced biosafety laboratory
Setting up a lab, even at this level, was not an easy task. Most of the equipment was monopolized by the United States and its sale to Iran is prohibited! One of the main equipment needed was handed over between 9 countries to reach Iran. In such a way that in each of these countries, the device was opened and registered once to register that country as a buyer. Upon the return of the manufacturer’s representative, the device is repackaged and shipped to the next country. In this way, after several shifts in the countries, the device was removed from the company’s coverage and the company’s representative did not go to the registry. Thus, it was possible to import this device to Iran with a delay of several months.
Another piece, which was needed for large-scale use on an industrial scale, was purchased by a few people so that everyone could buy only a limited number of them that could be justified in the laboratory. However, the same number was prevented from leaving the airport, and eventually less than half of these pieces arrived in Iran.
Breaking the monopoly of raw materials
Vaccine production, in addition to killing the virus, requires raw materials that are generally US-exclusive; From cell carriers to gels used in vaccines. Given the harsh US sanctions and the possibility of Iran joining the world of vaccinators, importing these substances was, if not impossible, at least extremely complicated. For example, in the case of a raw material, the police presented a scientific article to the buyer stating that this amount of raw material was more than what a laboratory needed, and in the end only allowed one-twentieth of it to leave. This incident showed the serious and comprehensive readiness of the Americans to prevent the sending of sensitive goods to Iran.
Problems of this kind caused the purchase of raw materials and at the same time the effort to make them to be immediately on the agenda. Some of these materials were purchased and others were localized by local knowledge-based companies. This major technological breakthrough greatly reduced Iran’s concerns about supplying raw materials on production days. At the same time, the productivity of production lines also increased. These developments did not remain at this point, and in the same vein, Iran achieved the possibility of an almost unlimited scale up in the production of the effective vaccine.

Iran’s access to knowledge increases the scale of production of the active ingredient of the vaccine
Which can use large bioreactors in the production of vaccines
Now the end point of the line
After producing the active ingredient, it was time to produce the vaccine. Most of Iran’s existing product lines were for a variety of children’s vaccines, producing a total of about 30 million doses a year. Not only could these lines not supply the 160 million Crohn’s vaccines needed by Iran, but it was basically impossible for them to go out of service, as children’s health vaccines also had to be produced daily and consumed by the community.
There was no choice; The Corona vaccine production line also had to be built from scratch. The first Barakat production line was launched based on the existing facilities in the country. The most important part of the production line is the “bioreactor” in which the reactions take place. A 500-liter bioreactor was the heart of the production line from a semi-functional vaccine production unit at the Barakat Institute. Two more 20- and 100-liter bioreactors were purchased from a factory and added to the production line. Relevant software was also updated with Iranian knowledge and Line 1 was put into operation in May 1400. The line produced about 8 million doses of vaccine in July and August, according to the plan, after the Barakat vaccine license was issued on June 15th.
The capacity of the first line was eventually 4 million doses of vaccine per month, and this number was far from what Iran needed. Although the informant, as the highest-ranking Blessing official, has repeatedly stressed the need to import vaccines over the past year, a threat called sanctions calls into question the possibility of doing so. So bigger units had to be built. Iranian companies were unable to produce and supply such equipment, and the purchase of vaccines from abroad also faced sanctions.
Development through India and China
Negotiations began with foreign companies. In December 1999, a Chinese company agreed to build a large unit in Iran with a production capacity of 25 million doses of vaccine per month in six months. This was the shortest time and most production capacity that Iran could achieve at that time. According to the plan, this line should be installed by July and entered the production cycle.
As time went on, American sanctions became more and more evident. In the winter of 1399, Iran had bought about 17 million doses of Kvax vaccine, 2 million doses of Bharat vaccine and 2 million doses of Sputnik vaccine, but by the end of 1999, less than 700,000 doses of vaccine reached Iran, about one third of which were gifts from China and Russia. Kwax did not give any vaccine and India only sent 125,000 doses of the vaccine and prevented the rest from being sent. Russian vaccines were also slowly being produced and imported.
The new year has arrived but the vaccine has not arrived. In April 1400, in order to get the vaccine to the people faster, Barakat bought another production line from India, which was to enter the production cycle in June. This line was smaller than the Chinese sample and with its 1200 liter bioreactor, it was possible to produce 8 million doses of vaccine per month.

US sanctions, manufacturers’ monopolies and the spread of new Corona strains
Caused the equipment of Barakat production line to arrive in Iran with a delay of several weeks
Corona complex tangle, monopoly and sanctions
The problem had just begun. An Indian company had begun manufacturing equipment that shut down the Indian Delta Corona. The equipment factory, like many in India, was shut down. After much negotiation, work finally resumed with a delay, but there was another problem. Flights between Iran and India airports were suspended and it was not possible to send equipment to Iran. After all these problems, the issue of sanctions has just arisen.
The United States had banned the sale of bioreactors to Iran on the pretext that the device was dual-purpose. The bioreactor, also known as Weslatank, arrived at the Indian airport through a foreign company. However, they did not allow the device to leave the airport and asked to talk to the buyer’s CEO. The paid bioreactor stayed at the airport for about a month, was not allowed to leave or return to the factory. A production line without a reactor is practically useless.
Eventually, this sensitive device was removed from the airport by removing the parts, and each part was sent to another country to come to Iran from there. In this route, some equipment was again confiscated by the countries for several days. With all the difficulties, the bioreactor parts and the production line finally arrived in Iran with about a month and a half delay and in July, as separate shipments.
The same thing happened with the Chinese production line; The United States stopped sending bioreactors, and the delivery of this device was delayed.
Of course, the sanctions were not limited to bioreactors. Most devices associated with the vaccine production line were identified and tracked by the United States. Because the use of these devices was clear and except Iran, only 6 other countries had the technology to use these devices. In addition to the usual sanctions, the economic dimension of the issue was also raised. In a word, the United States did not want the monopoly of American companies to produce vaccines to be broken and other countries to enter this market.

The Barakat vaccine production team has come under constant media attack in recent months
A strange but real problem
The devices, which were supposed to enter the production cycle in June and July, arrived in Iran with a delay of one to two months. The installation process of the production line was carried out quickly and it was still hoped that these production lines would be operational from the beginning of August and the promise of 50 million doses of vaccine would be fulfilled in September. But a new and strange problem emerged.
A group of Chinese and Indian engineers who had come to Iran to install devices and launch software all received coronas. All of these people claimed to have received the vaccine in their own country. Interestingly, the corona test of these people was negative at the beginning of their arrival in Iran, but immediately after 48 hours, the situation changed. Now it was blessed and the production line was almost installed but could not be used. There was no choice but to quarantine and host these people until they fully recover. A topic that lasted 2 to 3 weeks.
Finally, in late August, these people recovered and the final stages of setting up production lines began. The Indian production line was finalized in mid-September, increasing vaccine production by about 8 million doses per month. The Chinese production line is also in the final stages and will probably deliver its vaccines to the Iranian health network in less than a month.
These days, the Barakat vaccine has passed the three barriers of technological monopoly, Corona disease and US sanctions, but apparently, after facing all these strange events, Barakat officials do not intend to make any more promises about the amount of production. But it seems that if no other strange problem occurs, the promise of 50 million blessings will be fulfilled with a delay of about a month and a half.
The beginning of blessed days
The story of imports, however, is different. Three days after the Blessing Production License was issued, the United States, which practically no longer benefited from the vaccine embargo, officially lifted the Corona vaccine embargo in order to maintain at least its international prestige and to keep an eye on the Iranian market. This reduced the price of the imported vaccine from 15.5 euros to 8 euros, and the import of the vaccine increased exponentially from July. In July, as much as 5 months before that vaccine was introduced. Imports in August were twice as high as in July. The establishment of a new government intensified this process, and in September, more than 31.6 million doses of vaccine were imported. Thus, Iran, which had received only 6.8 million doses of vaccine in the five months before the embargo, has imported more than 51.4 million doses of vaccine in the three months since the blessing was produced.
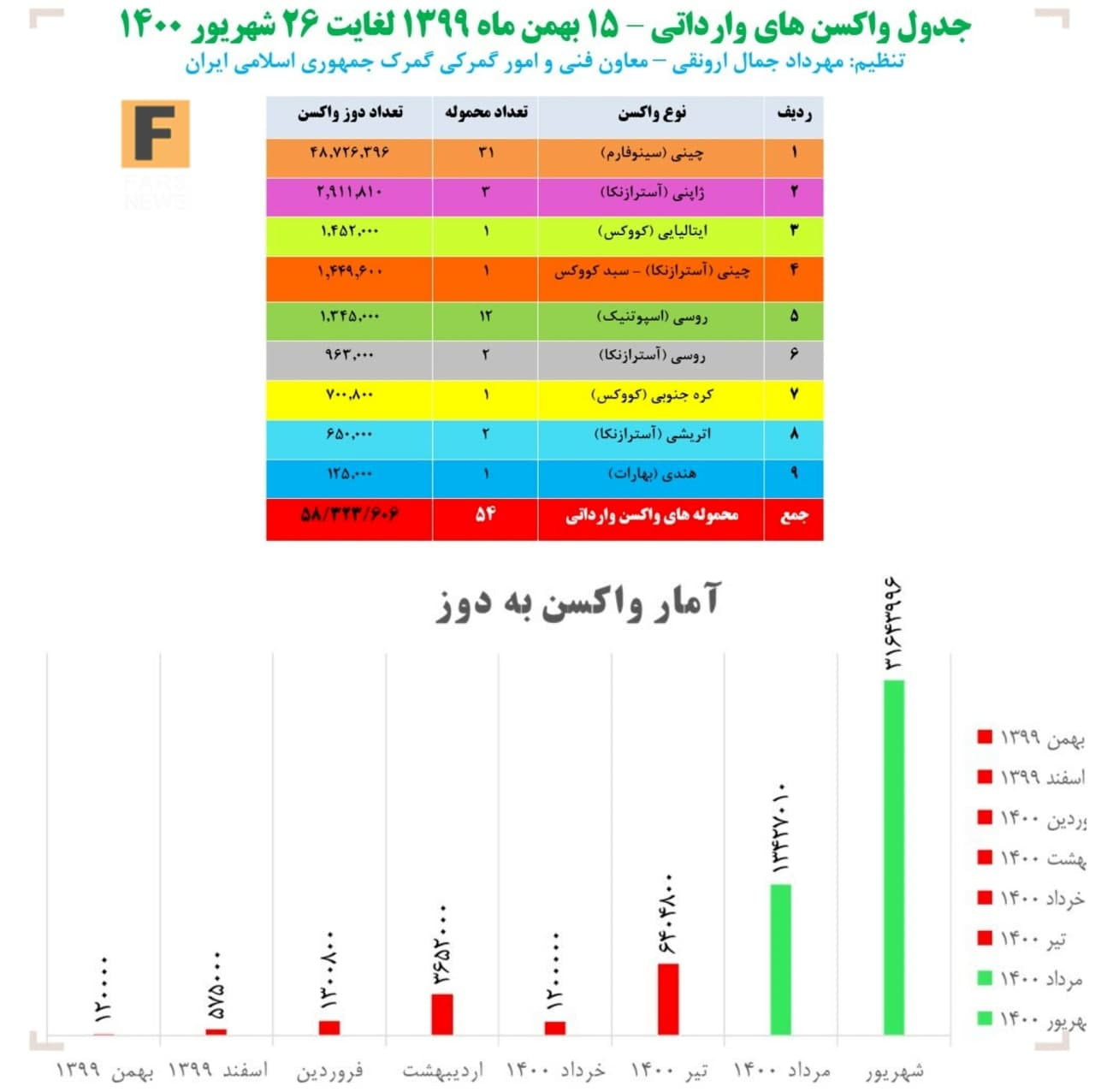
Barakat reduced the speed of vaccine imports by 12.5 times and halved its price.
End of message /
.

